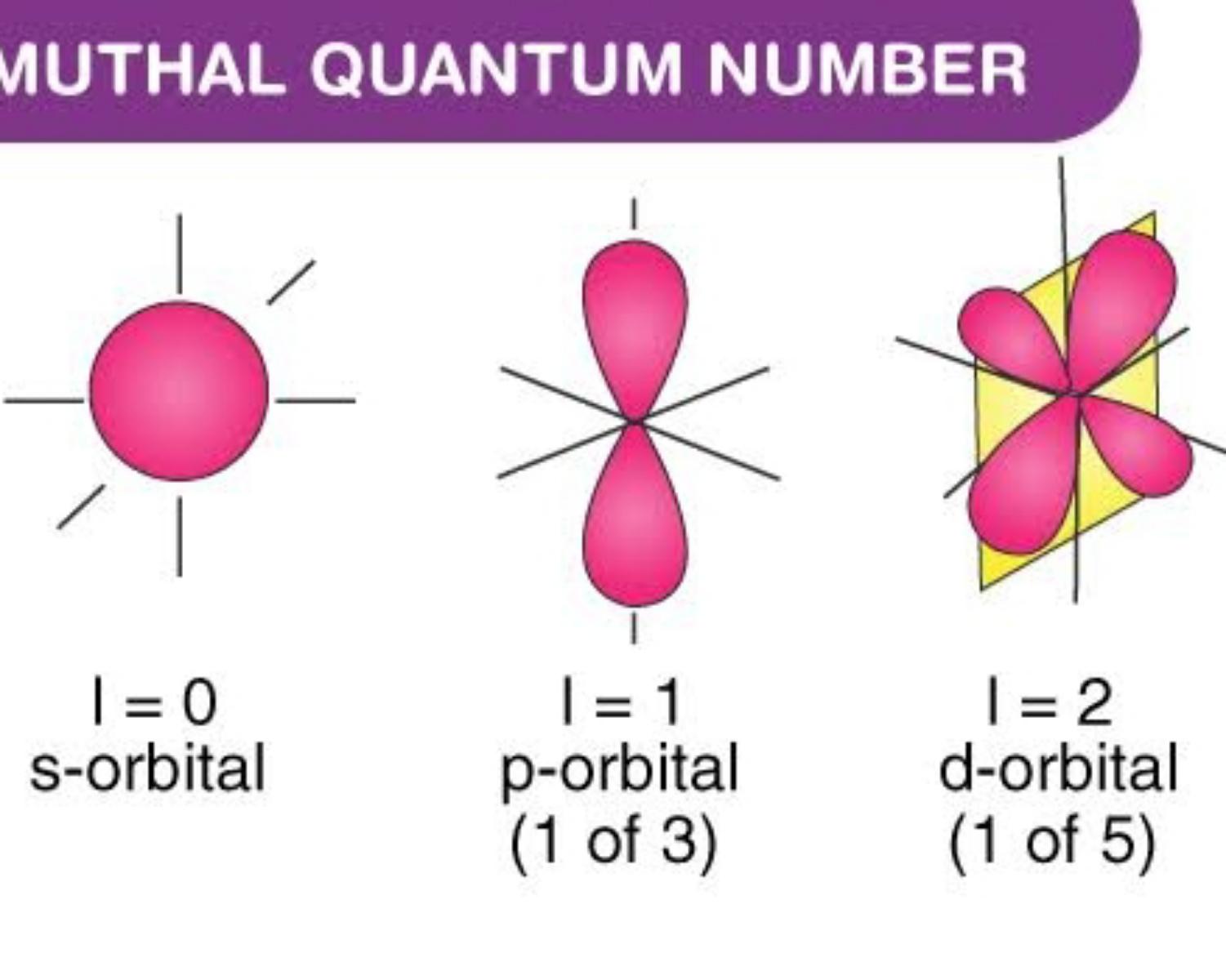
Azimuthal Quantum Numbers (ℓ)
The azimuthal quantum number is a quantum number for an atomic orbital that determines its orbital angular momentum and describes the shape of the orbital.
Every azimuthal number has his own capacity of electrons
- S(v=0) ==> 1 orb ==> 2 electrons
- P(v=1) ==> 3 orbs ==> 6 electrons
- D(v=2) ==> 5 orbs ==> 10 electrons
- F(v=3) ==> 10 orbs ==>12 electrons
Ps:open the photo to see the whole image. ((v = value))
photo owner: Byju's
25
65 reads
CURATED FROM
IDEAS CURATED BY
Highschool Student who has passionate interest in different sciences and World history.
The idea is part of this collection:
Learn more about scienceandnature with this collection
Basic survival skills
How to prioritize needs in survival situations
How to adapt to extreme situations
Related collections
Similar ideas to Azimuthal Quantum Numbers (ℓ)
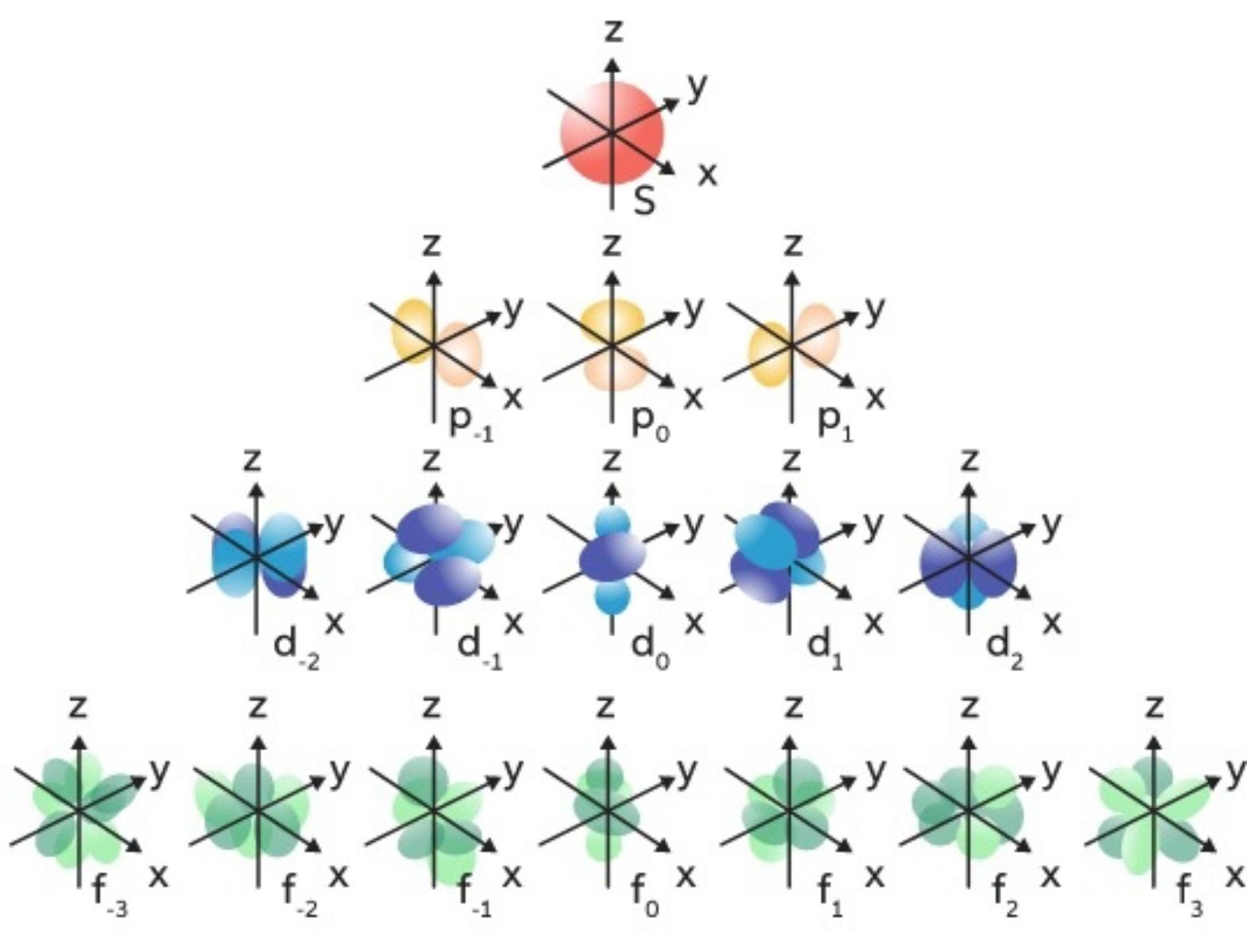
Magnetic quantum Numbers (mℓ)
Specifie the orientation in space of an orbital of a given energy (n) and shape (l). This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell.
Every orbital capacity is 2 electrons
Magnetic ...
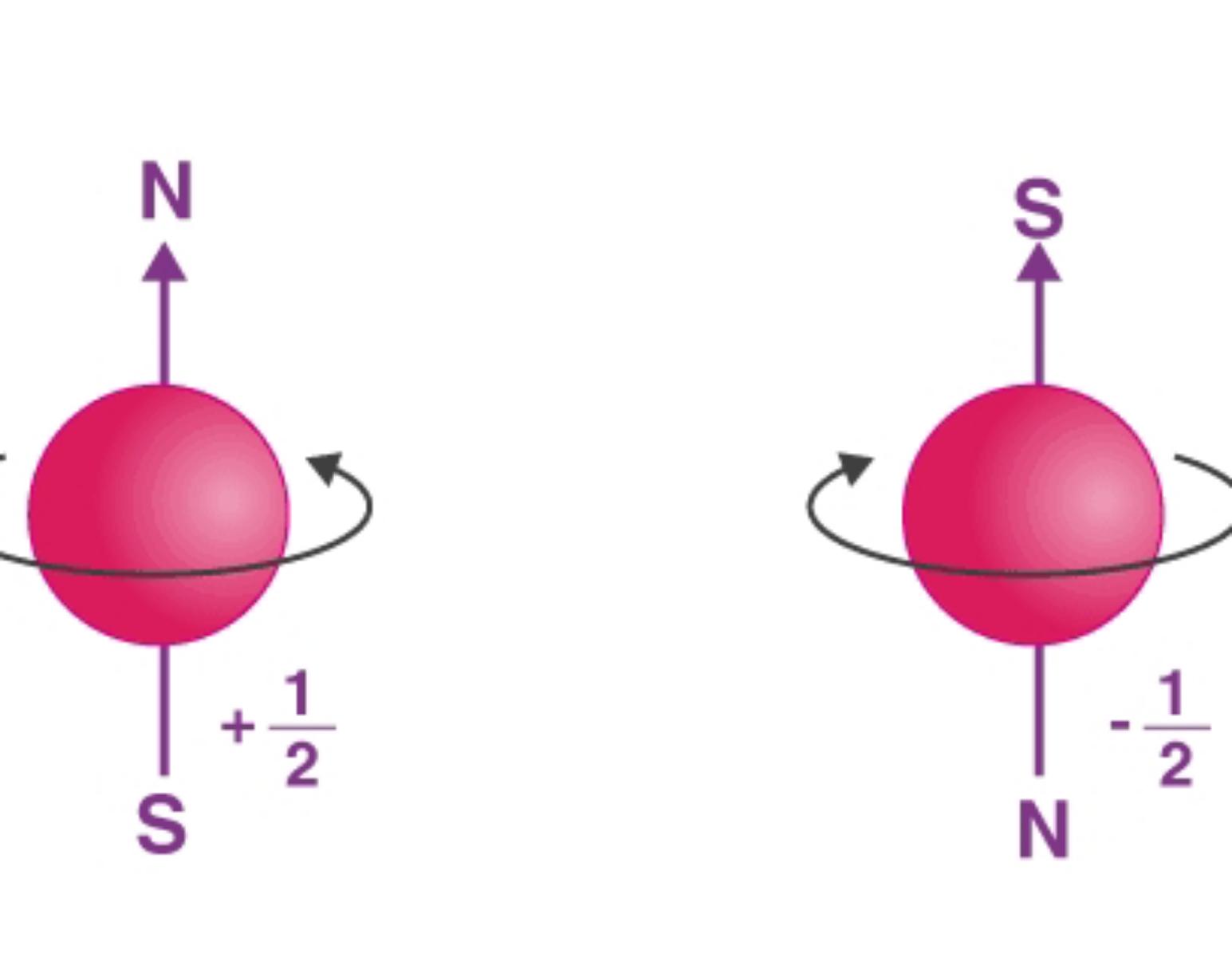
Spin Quantum Numbers (ms)
describe the angular momentum of an electron. An electron spins around an axis and has both angular momentum and orbital angular momentum. Because angular momentum is a vector, the Spin Quantum Number (s) has both a magnitude (½) and direction (+ or -)
(+½) ===> in the dire...
Practice
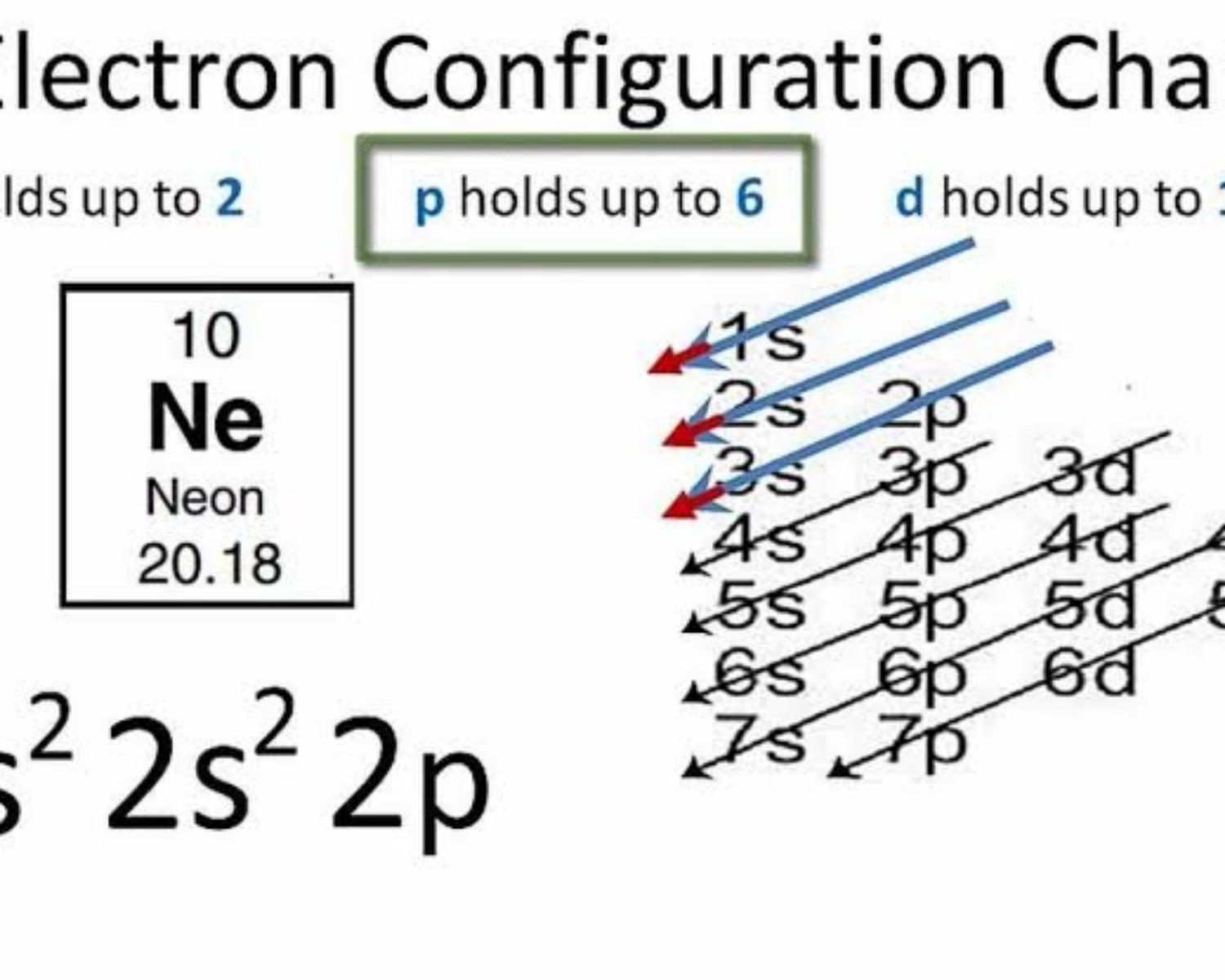
We have to define the exact place of the last electron for the element Neon¹⁰
- We have to configure the electrons 1s²,2s²,2p⁶
- Now we focus on the last number 2p⁶
• n (the number before the letter) =...
Read & Learn
20x Faster
without
deepstash
with
deepstash
with
deepstash
Personalized microlearning
—
100+ Learning Journeys
—
Access to 200,000+ ideas
—
Access to the mobile app
—
Unlimited idea saving
—
—
Unlimited history
—
—
Unlimited listening to ideas
—
—
Downloading & offline access
—
—
Supercharge your mind with one idea per day
Enter your email and spend 1 minute every day to learn something new.
I agree to receive email updates